Abstract
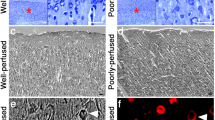
The loss of neuronal immunoreactivity of the cytoskeletal microtubule-associated protein 2 (MAP2) is known to be a marker of—at least—transient functional failure of neurons following ischemia. Because there are no specific neuropathological findings in forensic types of acute hypoxia-ischemia, detection of this relevant cause of death is often complicated and a reliable ischemic biomarker would be of great importance. We therefore investigated the neuronal immunoreactivity of MAP2 in human cases of forensic significance. A control group (n=27) was compared to a group of cases of hypoxia-ischemia (n=45), comprising death due to hanging (n=19), drowning (n=14) and carbon monoxide (CO) poisoning (n=12). Using immunohistochemical staining, the percentage of MAP2-positive neurons in the hippocampus (areas CA1–CA4) and frontal cortex (layers II–VI) was evaluated and compared. The hypoxia-ischemia group showed decreased MAP2 immunostaining in the hippocampal areas CA2–CA4 (P<0.05) and in cortical layers II–VI (P<0.001) compared to controls. Most vulnerable regions seem to be the hippocampal CA4 area and cortical layers III–V. Within the hypoxia-ischemia group, death due to CO poisoning was characterized by the lowest MAP2 immunoreactivity. The hypoxic-ischemic groups differ from controls by a distinct decrease of MAP2 immunostaining. Thus, the loss of MAP2 immunoreactivity may support the diagnosis of neuronal injury in forensic types of hypoxia-ischemia, although investigations on postmortem tissue must be interpreted cautiously.




Similar content being viewed by others
References
Akulinin VA, Dahlstrom A (2003) Quantitative analysis of MAP2 immunoreactivity in human neocortex of three patients surviving after brain ischemia. Neurochem Res 28:373–378
Auer RN (2000) Pure hypoxic and ischemic brain insults. In: Oehmichen M (ed) Brain hypoxia and ischemia. Research in legal medicine, 1st edn, vol 24. Schmidt-Römhild, Lübeck, pp 27–39
Auer RN, Siesjö BK (1988) Biological differences between ischemia, hypoglycemia, and epilepsy. Ann Neurol 24:699–707
Auer RN, Sutherland G (2002) Hypoxia and related conditions. In: Graham D, Lantos P (eds) Greenfield’s neuropathology, 7th edn, vol 1. Edward Arnold, London, pp 233–280
Bernaudin M, Nouvelot A, MacKenzie ET, Petit E (1998) Selective neuronal vulnerability and specific glial reactions in hippocampal and neocortical organotypic cultures submitted to ischemia. Exp Neurol 150:30–39
Brierly JB (1976) Cerebral hypoxia. In: Blackwood W, Corsellis JAN (eds) Greenfield’s neuropathology. Year Book Medical, Chicago, pp 43–85
Evers P, Uylings HBM (1994) Microwave-stimulated antigen retrieval is pH and temperature dependent. J Histochem Cytochem 42:1555–1563
Evers P, Uylings HBM (1997) An optimal antigen retrieval method suitable for different antibodies on human brain tissue stored for several years in formaldehyde fixative. J Neurosci Methods 72:197–207
Grady MS, Charleston JS, Maris D, Witgen BM, Lifshitz J (2003) Neuronal and glial cell number in the hippocampus after experimental traumatic brain injury: analysis by stereological estimation. J Neurotrauma 20:929–941
Hirokawa N (1994) Microtubule organization and dynamics dependent on microtubule-associated proteins. Curr Opin Cell Biol 6:74–81
Hossmann KA (2000) Pathophysiology of brain resuscitation after cardiac arrest. In: Oehmichen M (ed) Brain hypoxia and ischemia. Research in legal medicine, 1st edn, vol 24. Schmidt-Römhild, Lübeck, pp 145–164
Huh JW, Raghupathi R, Laurer HL, Helfaer MA, Saatman KE (2003) Transient loss of microtubule-associated protein 2 immunoreactivity after moderate brain injury in mice. J Neurotrauma 20:975–984
Inamura K, Olsson Y, Siesjö BK (1987) Substantia nigra damage induced by ischemia in hyperglycemic rats. A light and electron microscopic study. Acta Neuropathol (Berl) 75:131–139
Irving EA, McCulloch J, Dewar D (1997) The effect of postmortem delay on the distribution of microtubule-associated proteins τ, MAP2, and MAP5 in the rat. Mol Chem Neuropathol 30:253–271
Ito U, Spatz M, Walker JT Jr, Klatzo I (1975) Experimental cerebral ischemia in mongolian gerbils. I. Light microscopic observations. Acta Neuropathol (Berl) 32:209–223
Jenkins LW, Povlishock JT, Lewelt W, Miller JD, Becker DP (1981) The role of postischemic recirculation in the development of ischemic neuronal injury following complete cerebral ischemia. Acta Neuropathol (Berl) 55:205–220
Johnson GVW, Jope RS (1992) The role of microtubule-associated protein 2 (MAP-2) in neuronal growth, plasticity, and degeneration. J Neurosci Res 33:505–512
Kalimo H, Olsson Y, Paljärvi L, Söderfeldt B (1982) Structural changes in brain tissue under hypoxic-ischemic conditions. J Cereb Blood Flow Metab 2:19–22
Kirino T (1982) Delayed neuronal death in the gerbil hippocampus following ischemia. Brain Res 239:57–69
Kirino T, Sano K (1984) Selective vulnerability in the gerbil hippocampus following transient ischemia. Acta Neuropathol (Berl) 62:201–208
Kitagawa K, Matsumoto M, Niinobe M, Mikoshiba K, Hata R, Ueda H, Handa N, Fukunaga R, Isaka Y, Kimura K, Kamada T (1989) Microtubule-associated protein 2 as a sensitive marker for cerebral ischemic damage—immunohistochemical investigation of dendritic damage. Neuroscience 31:401–411
Kitamura O, Gotohda T, Ishigami A, Tokunaga I, Kubo S, Nakasono I (2005) Effect of hypothermia on postmortem alterations in MAP2 immunostaining in the human hippocampus. Leg Med (Tokyo) 7:24–30
Knight B (2004) Forensic pathology, 3rd edn. Arnold, London
Kwei S, Jiang C, Haddad GG (1993) Acute anoxia-induced alterations in MAP2 immunoreactivity and neuronal morphology in rat hippocampus. Brain Res 620:203–210
Leifer D, Kowall NW (1993) Immunohistochemical patterns of selective cellular vulnerability in human cerebral ischemia. J Neurol Sci 119:217–228
Lipton P (1999) Ischemic cell death in brain neurons. Physiol Rev 79:1431–1568
Malinak C, Silverstein FS (1996) Hypoxic-ischemic injury acutely disrupts microtubule-associated protein 2 immunostaining in neonatal rat brain. Biol Neonate 69:257–267
Oehmichen M (1990) Neuropathologie der forensisch relevanten Formen des Erstickens. In: Brinkmann B, Püschel K (eds) Ersticken. Fortschritte in der Beweisführung, 1st edn. Springer, Berlin, pp 151–157
Oehmichen M, Meissner C (2000) Forensic neuropathological aspects of cerebral anoxia/ischemia and hypoxia/hypoxemia. In: Oehmichen M (ed) Brain hypoxia and ischemia. Research in legal medicine, 1st edn, vol 24. Schmidt-Römhild, Lübeck, pp 13–25
Oehmichen M, Meissner C, Wurmb-Schwark N von, Schwark T (2003) Methodical approach to brain hypoxia/ischemia as a fundamental problem in forensic neuropathology. Leg Med (Tokyo) 5:190–201
Oehmichen M, Auer RN, König HG (2006) Forensic Neuropathology and associated neurology. Springer, Berlin Heidelberg New York
Ota A, Ikeda T, Ikenoue T, Toshimori K (1997) Sequence of neuronal responses assessed by immunohistochemistry in the newborn rat brain after hypoxia-ischemia. Am J Obstet Gynecol 177:519–526
Petito CK, Pulsinelli WA (1984) Delayed neuronal recovery and neuronal death in rat hippocampus following severe cerebral ischemia: possible relationship to abnormalities in neuronal processes. J Cereb Blood Flow Metab 4:194–205
Piantadosi CA, Zhang J, Demchenko IT (1997) Production of hydroxyl radical in the hippocampus after CO hypoxia or hypoxic hypoxia in the rat. Free Radic Biol Med 22:725–732
Qi JP, Wu AP, Wang DS, Wang LF, Li SX, Xu FL (2004) Correlation between neuronal injury and caspase-3 after focal ischemia in human hippocampus. Chin Med J (Engl) 117:1507–1512
Schwab C, Bondada V, Sparks DL, Cahan LD, Geddes JW (1994) Postmortem changes in the levels and localization of microtubule-associated proteins (tau, MAP2 and MAP1B) in the rat and human hippocampus. Hippocampus 4:210–225
Shi SR, Key ME, Kalra KL (1991) Antigen retrieval in formalin-fixed, paraffin-embedded tissues: an enhancement method for immunohistochemical staining based on microwave oven heating of tissue sections. J Histochem Cytochem 39:741–748
Shi SR, Chaiwun B, Young L, Cote RJ, Taylor CR (1993) Antigen retrieval technique utilizing citrate buffer or urea solution for immunohistochemical demonstration of androgen receptor in formalin-fixed paraffin sections. J Histochem Cytochem 41:1599–1604
Shi SR, Cote RJ, Taylor CR (2001) Antigen retrieval techniques: current perspectives. J Histochem Cytochem 49:931–937
Simon RP (1999) Hypoxia versus ischemia. Neurology 52:7–8
Smith ML (2000) Injury-determining factors and cellular mechanisms for ischemic damage in the brain. In: Oehmichen M (ed) Brain hypoxia and ischemia. Research in legal medicine, 1st edn, vol 24. Schmidt-Römhild, Lübeck, pp 59–64
Suurmeijer AJH, Boon ME (1993) Notes on the application of microwaves for antigen retrieval in paraffin and plastic tissue sections. Eur J Morphol 31:144–150
Tomimoto H, Yanagihara T (2000) Vulnerability of dendrites and nerve cell bodies in cerebral ischemia. In: Oehmichen M (ed) Brain hypoxia and ischemia. Research in legal medicine, 1st edn, vol 24. Schmidt-Römhild, Lübeck, pp 65–84
Torvik A (1984) The pathogenesis of watershed infarcts in the brain. Stroke 15:221–223
Trojanowski JQ, Schuck T, Schmidt ML, Lee VMY (1989) Distribution of phosphate-independent MAP2 epitopes revealed with monoclonal antibodies in microwave-denatured human nervous system tissues. J Neurosci Methods 29:171–180
Tucker RP (1990) The roles of microtubule-associated proteins in brain morphogenesis: a review. Brain Res Brain Res Rev 15:101–120
Vanicky I, Balchen T, Diemer NH (1995) Alterations in MAP2 immunostainability after prolonged complete brain ischaemia in the rat. Neuroreport 7:161–164
Yamamoto K, Morimoto K, Yanagihara T (1986) Cerebral ischemia in the gerbil: transmission electron microscopic and immunoelectron microscopic investigation. Brain Res 384:1–10
Yamaoka Y, Shimohama S, Kimura J, Fukunaga R, Taniguchi T (1993) Neuronal damage in the rat hippocampus induced by in vivo hypoxia. Exp Toxicol Pathol 45:205–209
Yanagihara T, Brengman JM, Mushynski WE (1990) Differential vulnerability of microtubule components in cerebral ischemia. Acta Neuropathol 80:499–505
Zhang J, Piantadosi CA (1992) Mitochondrial oxidative stress after carbon monoxide hypoxia in the rat brain. J Clin Invest 90:1193–1199
Acknowledgements
We wish to thank Susanne Rath for her kind assistance with tissue preparation as well as Silke Tychsen-Langer for excellent secretarial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kühn, J., Meissner, C. & Oehmichen, M. Microtubule-associated protein 2 (MAP2)—a promising approach to diagnosis of forensic types of hypoxia-ischemia. Acta Neuropathol 110, 579–586 (2005). https://doi.org/10.1007/s00401-005-1090-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-005-1090-9